Rapid Atp bioluminescence methods must be able to overcome different technical challenges to be reliable.
To understand the technical challenges faced by conventionnal Atp test methods, it is necessary to understand that there are different kinds of Atp bioluminescence assays.
Classic methods Atp bioluminescence test kits available on the market are divided in two different categories,
Each of these Atp bioluminescence assay have their own limitations.
But let see first the difference between these two different types of ATP bioluminescence test kits, to understand the issues involved.
Conventional 1st, 2nd generation ATP bioluminescence test kits
First generation Atp bioluminescence refers to total Atp bioluminescence method where free Atp molecules and intracellular Atp molecules are analyzed together without any discrimination.
Second generation Atp bioluminescence refers to ATP methods quantifiyng only intracellular ATP without any discrimination of the cell type, whether this a plant cell, somatic cell, or microorganisms cell.

Before going into investigating the issues that 1st and 2nd generation Atp bioluminescence assays do not solve, it is usefull to understand what different molecules and cells in samples impact Atp bioluminescence measured.
Which molecules & cells in sample affect Atp bioluminescence?
Classic ATP (adenosine triphosphate) bioluminescence methods encounters many unsolved technical issues. This makes their ATP tests results unreliable. Let’s see why.
Issues unsolved by 1st & 2nd generation Atp test kits..
Inhibiting substances triggers false negatives
Free ATP triggers false positives
Molecules present in beverages, and other liquids, if not removed (sulfur dioxide, metal ions, tannins, polyphenols) inhibit Atpbioluminescence reaction.
Swabbing collects on surface traces of detergents who are freed into the luminometer test tube and released into the luciferin-luficerase.
Consequently, these inhbiting substances will affect the luciferin-luciferase reaction.
This can be even to the point of reducing considerably the Atp bioluminescence reaction measured by the Atp-meter or luminometer. The lower luminescent values measured because of this quenching effect, will give false negative results.
These false negatives will make the user to believe that the microbial population in the sample is low or non-existent, whereas it is not the case!
Beverages like beer contain lots of free ATP of plant origin released during brewing.
When yeasts multiply, they bud and produce daughter cells. These daughter yeast cells detach from the mother cell, and release huge amounts of ATP.
If this free ATP is analyzed together with microorganisms it triggers false positives.
These false positives suggest that sample analyzed contains many microorganisms, meanwhile bioluminescence values are mainly due to free Atp..
Is there a correlation between ATP & concentration of viable microorganisms?

Theoretically, higher the amount of ATP released, higher the intensity of ATP bioluminescence in the presence of luciferin-luciferase would be, higher the number of viable microorganisms present in the sample would also be.
1st & 2nde generation ATP test kits are not specific.
In a mixture of different cells of various origins, the classical methods of measuring Atp bioluminescence, whether 1st or 2nd generation, are unable to discriminate microbial Atp from the ATP of other plant or somatic cells, for example during surface sampling.
Reagents used generally lyse all cells without discrimination of cell types, and release ATP from all cells. This is particularly problematic because some non-microbial cells contain huge amounts of ATP and therefore the bioluminescence values by this non-microbial Atp will never allow to quantify the microorganism population.
3rd generation ATP bioluminescence kit solves these issues
Third generation ATP test kits refers to method quantifying microbial intracellular ATP discriminated from somatic cells ATP.
This method uses a filter-cuvette combination with specific somatic releasing agent and a microbial lysing reagent and was successfully used and published for water, surface, air and meat carcasses atp bioluminescence studies.
3rd generation Atp bioluminescence assay was originally named Profile-1 and invented by New Horizons Diagnostics Inc. in Baltimore.
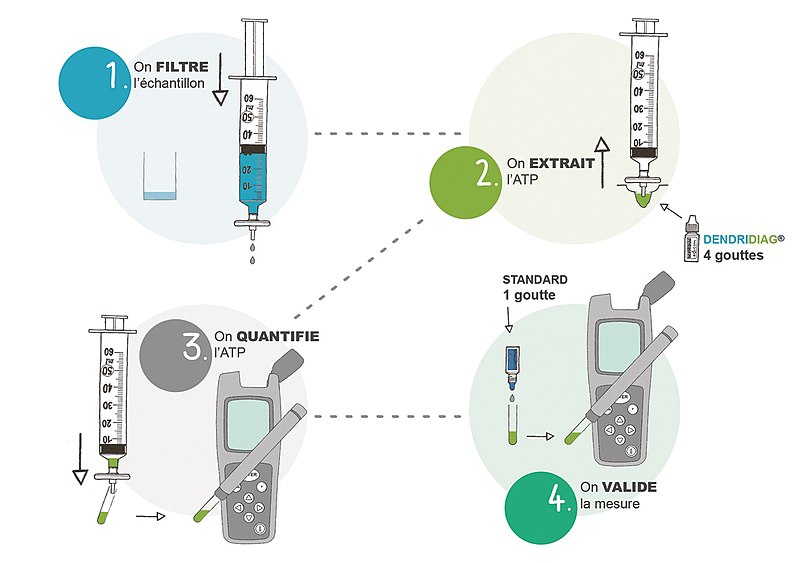
It is allow to concentrate cells with a syringe and a Cell concentrator containing a removable cuvette-filter the Filtravette™.
This Filtravette invention allowed to concentrate, flush inhibiting substances, and make different sample treatment and trigger and quantify Atp bioluminescence reaction within the same cuvette.




Here on the right two graphs showhow Filtravette wash efficiency with Somatic releasing agent flush TCA extractant and Bleach to avoid quenching of bioluminescence signal.


3rd generation Atp bioluminescence Profile-1 test kit, due to its reliability and resistance to quenching substances has been very often published in numerous independant correlation studies. These studies validated the correlation between plate count methods and Atp bioluminescence values in a sample detected by Profile-1.
Numerous correlation studies with 3rd generation Atp test kits



Select the correlation studies published by clicking on the different buttons.
However 3rd generation ATP test kits have their limits, they are not discriminating selectively microorganisms for other non-targeted microorganisms.
3rd generation test kit limits: unable to selectively discriminate yeast from bacteria.
Therefore conventional ATP test methods, whether 1st, 2nd ir 3rd generation Atp bioluminescence methods, cannot differentiate yeasts from bacteria, when yeast ATP is analyzed together with bacterial ATP.
Yeasts form very often clusters with bacteria, which are very difficult , if not impossible, to separate by filtration or centrifugation.
This is one of the reasons why these classic Atp bioluminescence methods are not used in oenology except for hygiene control, as a single yeast cell contains as much ATP as 100 viable bacterial cells.
On the other hand, important populations of lactic acid bacteria (10 exponential 7 / ml) are found during wine aging. These bacteria may overwhelm bioluminescence results making it impossible to determine number of viable yeasts in the sample.
Does it mean that there is no solution to these unsolved issues.? Indeed there is a solution for: Rapid selective Atp bioluminescence discrimating yeast from certain bacteria.
Click on below button to see the solution.
